Tags

CHESS celebrates construction milestone with Wilson West open house
On Wednesday, November 15, CHESS had an open house for members of the Cornell community in the new Wilson West expansion project. The project recently received its temporary certificate of occupancy, which marks a milestone in the construction project.

CHESS user Ando wins award for contributions to biochemistry and molecular biology
Nozomi Ando, associate professor of chemistry and chemical biology, is being honored with the 2024 American Society for Biochemistry and Molecular Biology (ASBMB) Mildred Cohn Young Investigator Award for her outstanding and field defining contributions to the fields of protein dynamics.

Gabrielle Illava: From SERCCS to PhD
We met with Gabrielle Illava to talk about her journey in the academic research world. Gabrielle started in the CHESS program: Summer Engineering and Research for Community College Students (SERCCS). She also transferred into Cornell from the nearby Tompkins Cortland Community College (TC3) to finish her undergraduate degree in Molecular and Cell Biology, before pursuing her PhD at CLASSE, which she completed in the spring of 2023.

CHESS Research Uses Pressure To Understand RNA Dynamics
High pressure induces excited states, suggests role in HIV infection

Inline small-angle X-ray scattering-coupled chromatography under extreme hydrostatic pressure
A new paper appearing in the journal “Protein Science” and authored by CHEXS-supported graduate student Robert Miller demonstrates for the first time that reproducible chromatographic separations coupled directly to high-pressure BioSAXS can be achieved at pressures up to at least 100 MPa.
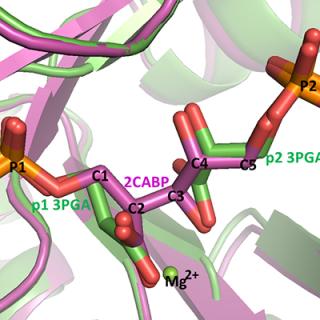
Crystal structure of a type III Rubisco in complex with its product 3-phosphoglycerate
Recent research performed at CHESS gives insight into intermediate stages of Rubisco’s catalysis mechanism.
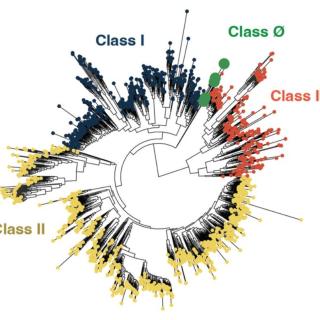
Protein family shows how life adapted to oxygen
Cornell scientists have created an evolutionary model that connects organisms living in today’s oxygen-rich atmosphere to a time, billions of years ago, when Earth’s atmosphere had little oxygen.
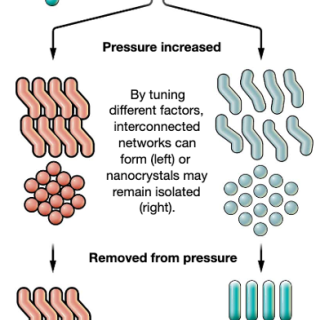
Nanocrystals with Metastable High-Pressure Phases Under Ambient Conditions
This groundbreaking work was supported by a collaboration between the PI an collaborators and CHEXS scientist Zhongwu Wang, spanning many years, across the former CHESS “B-line” beamline and the new CHEXS HPBio facilities. This work was enabled by custom diamond anvil cells with large opening angles for simultaneous SAXS / WAXS measurements, and by in-situ spectroscopy techniques deployed at CHESS





