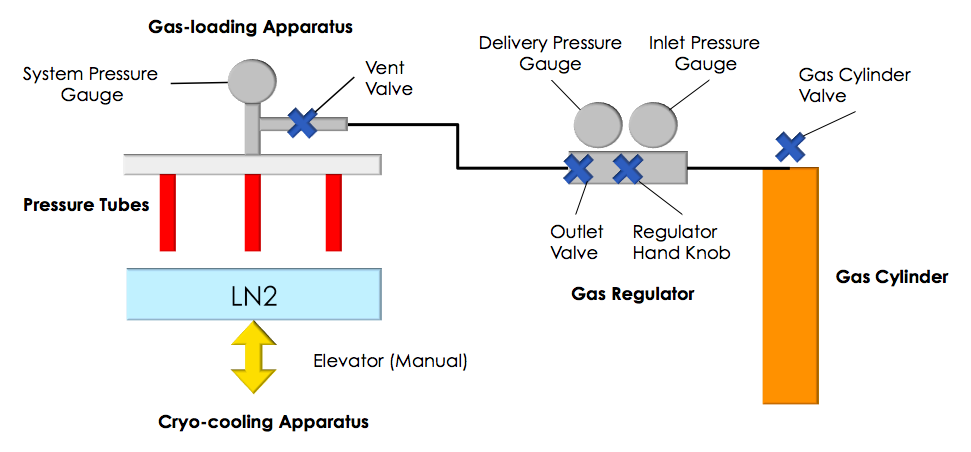
High-pressure cryocooling was developed primarily for crystal cryoprotection with minimal damage due to the cryocooling process, and dozens of unique macromolecular crystals have been successfully cryopreserved, including membrane protein crystals (e.g. a Kv ion channel; cytochrome c oxidase) and large protein/protein and RNA/protein complexes, such as the ribosome. Read more...
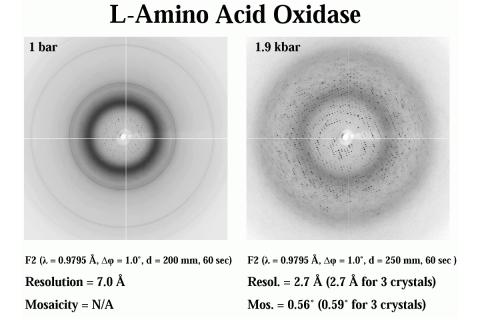
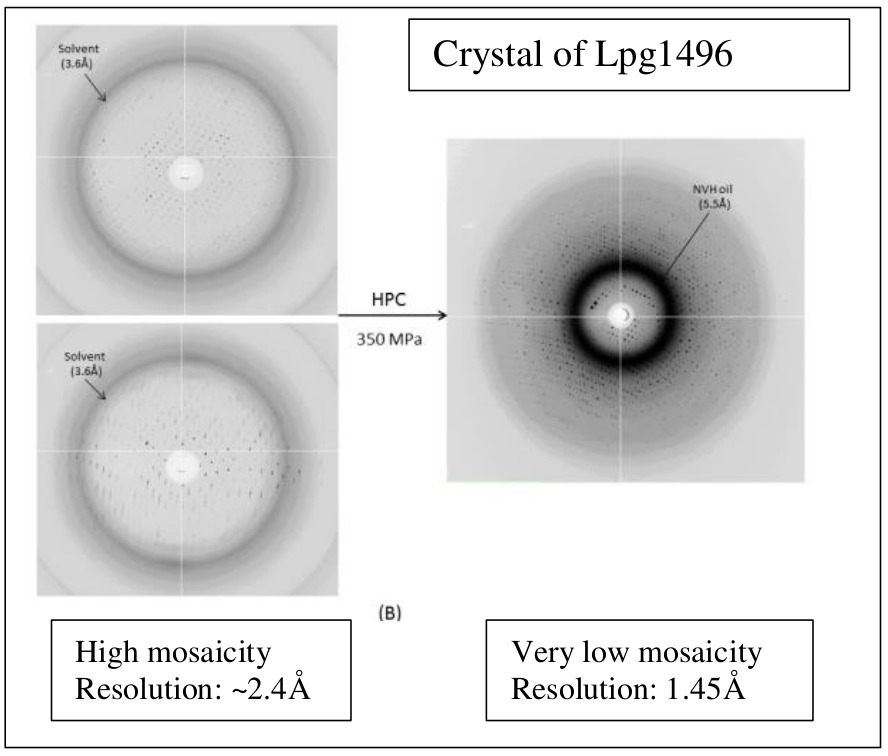
It was originally thought that HPC could not improve the as-grown quality of a crystal, but it turns out that this is not true in all cases. HPC has been used to reduce mosaicity as well as anisotropy of diffraction, improve the resolution, and even remove some non-merohedral twinning. Read more...
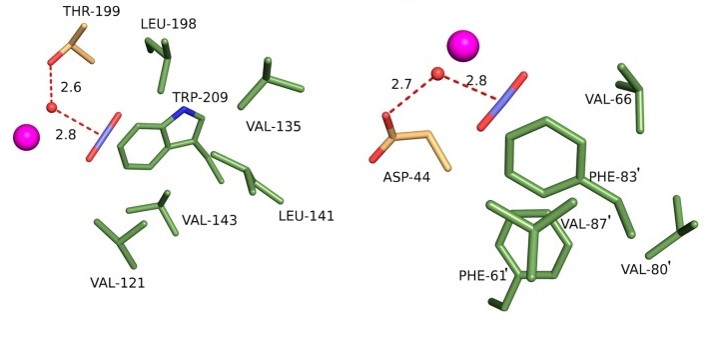
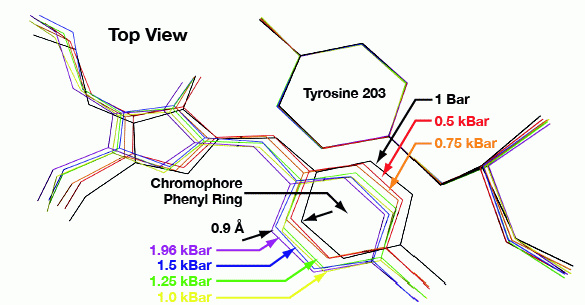
HPC has also been successfully applied to stabilize ligand–protein interactions and to study the pressure effect on the structure of proteins (such as yellow fluorescent protein and citrine). Morover, it has been used to study the phase behavior of water inside protein crystals and its relationship with protein dynamics at cryogenic temperatures. The method has also been extended to anomalous diffraction phasing by incorporating noble gases such as Kr and Xe. Read more...
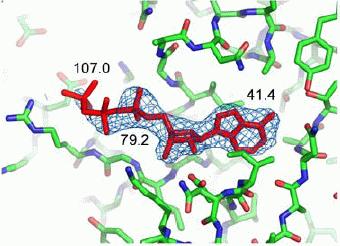
Furthermore, the procedure has been modified to entrap gas molecules, e.g. carbon dioxide in carbonic anhydrase, in order to visualize intermediate states in the functioning of enzymes. Read more...