Sidebar Menu (View Pages)
- Status
- ⌃ Science
- ⌃ Users
- ⌃ Facilities
- ⌃ Public
- Industry
- ⌃ About
Tags
Featured

Unlocking the Mysteries of Life Under Pressure
As scientists continue to discover new niches for extreme life, the biological relevance of hydrostatic pressure is becoming much more widely understood and appreciated. The unusual adaptations of organisms thriving under these conditions promise to be a rich source of new insights, provided structural information can be obtained at the molecular level.
CHESS is at the forefront of this research - enabling scientists to study samples under high pressure, revealing how biomolecules and cellular structures behave in extreme environments.

Cultivating Community at the 2024 CHESS Annual User Meeting
CHESS celebrated its annual User Meeting with the theme "Cultivating Community," emphasizing the integral role of CHESS users, researchers, technical staff, and scientific staff.
The 2024 User Meeting attracted over 150 attendees who gathered for a day of scientific talks, facility updates, awards, and a vibrant poster session. See the Photo Gallery here!
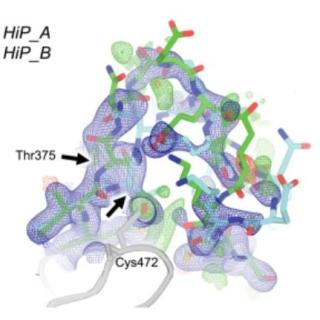
Pushed to extremes: distinct effects of high temperature versus pressure on the structure of STEP
In a new paper appearing in Communications Biology, prof. Daniel Keedy's group from the CUNY Advanced Science Research Center used x-ray crystallography at CHESS to investigate the conformation changes of a medically-important biomolecule (the protein tyrosine phosphatase enzyme STEP) when perturbed by pressure and temperature.

Summer 2024 Undergraduate Students at CHESS
The Cornell Laboratory for Accelerator-based Sciences and Education (CLASSE) funds summer research opportunities in the areas of x-ray and accelerator sciences, materials science, chemistry, and mechanical engineering for pre-selected undergraduate students from primarily undergraduate institutions and minority serving institutions. This summer, twenty REU students are hosted by CLASSE. Ten students from the CHESS summer programs -- SERCCS, PREM, and SUNRiSE -- are jumping into their rigorous summer undergraduate research experience.

Reflections: 2024 Wilson Synchrotron Laboratory Open House
The Open House was a chance for science enthusiasts of all ages to come and see what we do behind the scenes at CHESS and CLASSE! This event was first and foremost about community, from our staff and students, to the local Ithaca and Cornell communities – we hope to inspire dozens of young new scientists!

Wilson West Update - January 2024
The Wilson West construction project is about to enter the next phase as we head into construction and installation of interior X-ray facilities.

CHESS celebrates construction milestone with Wilson West open house
On Wednesday, November 15, CHESS had an open house for members of the Cornell community in the new Wilson West expansion project. The project recently received its temporary certificate of occupancy, which marks a milestone in the construction project.

CHESS user Ando wins award for contributions to biochemistry and molecular biology
Nozomi Ando, associate professor of chemistry and chemical biology, is being honored with the 2024 American Society for Biochemistry and Molecular Biology (ASBMB) Mildred Cohn Young Investigator Award for her outstanding and field defining contributions to the fields of protein dynamics.





