Trifluoromethyl (CF3) substituents profoundly influence properties of organic molecules and transition metal complexes. Medicinal chemists have recognized the value of the CF3 group for advantageous drug properties including increased physiological longevity, facile blood−brain barrier penetration, and enhanced protein−substrate binding affinities. Consequently, developing reactions that install CF3 on organic frameworks is a major research goal.
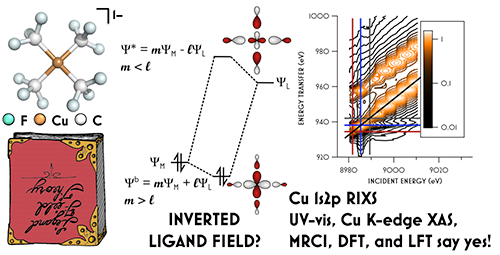
These data, supported by density functional theory, multiplet theory, and multireference calculations, support a surprising ground state electronic configuration with lowest unoccupied orbital predominantly of trifluoromethyl character. The consensus 3d10 configuration features an inverted ligand field in which all five metal-localized molecular orbitals are located at lower energy relative to the trifluoromethyl-centered σ orbitals.
Understanding the electronic structures of transition metal catalysts and reagents is key to controlling their reactivity. The present study sets the stage for development of new trifluoromethylating agents based on earth-abundant Cu that exploit electrophilic rather than nucleophilic reactivity.
Reference:
Walroth RC, Lukens JT, MacMillan SN, Finkelstein KD, and Lancaster KM, "Spectroscopic Evidence for a 3d10 Ground State Electronic Configuration and Ligand Field Inversion in [Cu(CF3)4]1−"
![RIXS spectrum of [Cu(CF)]](/sites/default/files/styles/large/public/2018-02/toc-fixed_graphic.jpg?itok=hyGLYu9T)