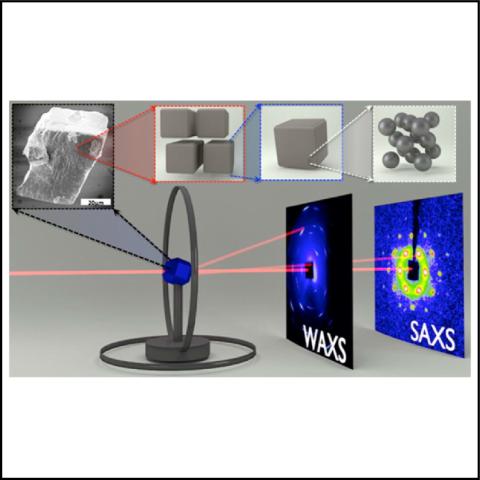
What did the Scientists Discover?
Nanocrystals are perfect periodic arrangements of atoms that only extend a few tens of nanometers in any direction and exhibit unique electronic, optic and/or magnetic characteristics determined by their nanoscale dimensions. Assembling nanocrystals into two or three dimensional ordered configurations results so called superlattices or supercrystals. The characteristics of these superstructures reflect the unique physical and chemical properties of the nanocrystal building blocks as well as the impact of strong delocalization and electronic coupling through well-arranged and embedded interfaces throughout the superstructure.
In this review, CHESS researchers summarize their findings on superstructures consisting of different types of nanocrystals (nanoparticle shape, lattice symmetry, composition) and explain the symmetry and characteristics of the resulting superstructures.
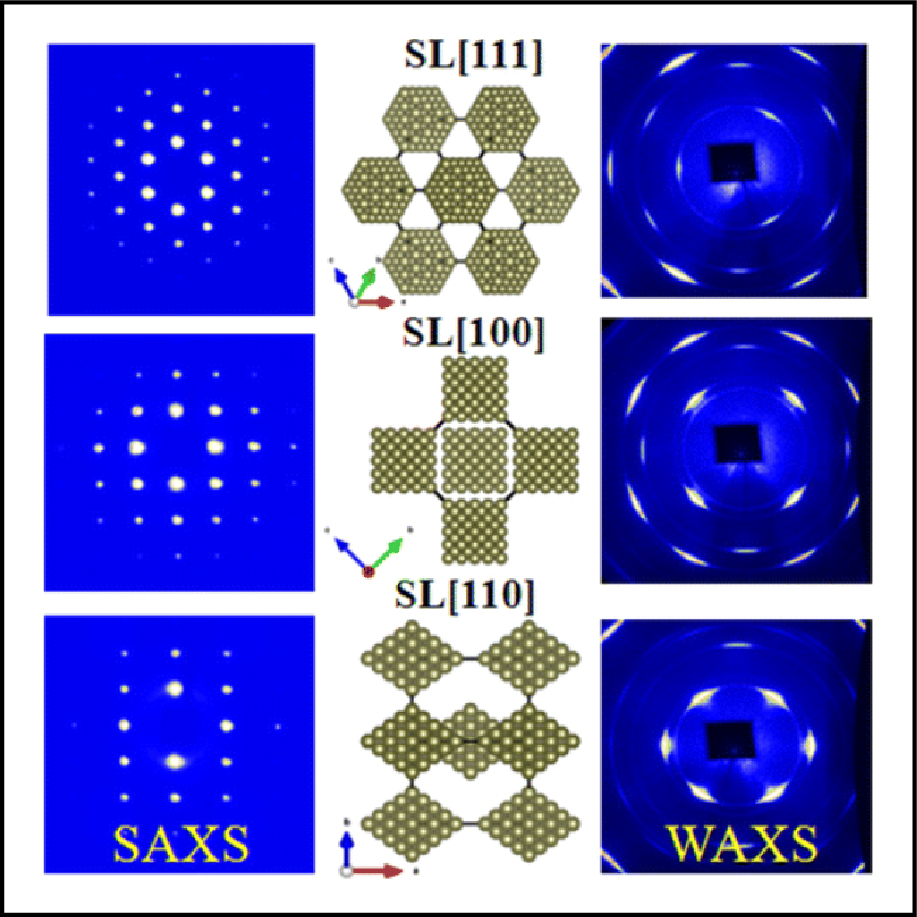
What are the broader impacts of this work?
Based on this work, researchers can now predict how experimental conditions need to be configured to precisely control the self-assembly of nanocrystals into well ordered superlattices with specifically designed atomically crystallographic orientations and translational symmetry resulting in desired materials properties.
Why did this research need CHESS?
Both small- and wide-angle X-ray scattering (SAXS/WAXS) data were simultaneously measured from the same volume of a superstructure at CHESS. The careful analysis of the images provided reliable structural information and correlations across multiple length scales crucial for understanding nanocrystal superlattices.
Collaborators:
- Xin Huang, Cornell High Energy Synchrotron Source, Wilson Laboratory, Cornell University
- Zhongwu Wang, Cornell High Energy Synchrotron Source, Wilson Laboratory, Cornell University
Publication Citation:
Huang, X.; Wang, Z., “Supercrystallography-Based Decoding of Structure and Driving Force of Nanocrystal Assembly,” Materials, 2019, 12, 3771.https://doi.org/10.3390/ma12223771
How was the work funded?
CHESS was supported by NSF Grant DMR-1332208. Moreover, financial support from the Center for Alkaline-Based Energy Solutions (CABES), funded by the U.S. Department of Energy under Award No. DE-SC0019445 is acknowledged.