What did the Scientists Discover?
Enzymes are Nature's catalysts, essential for carrying out countless life processes including everything from water splitting to DNA synthesis. These attributes make enzymes attractive molecules to study, both for learning new ways to catalyze chemical reactions and also for discovering new targets for pharmaceuticals. These goals, however, require us to know not only what reaction an enzyme catalyzes, but also how it carries out that reaction. One particularly interesting enzyme is a ribonucleotide reductase (RNR) from the human Chlamydia pathogen. This RNR is responsible for producing the building blocks of DNA for the pathogen and is not present in humans, making it an attractive drug target for future treatments. Unfortunately, important details about how this enzyme works are still unknown, a fact which motivated the collaboration between teams at Penn State and CHESS to investigate the mechanism of this RNR.
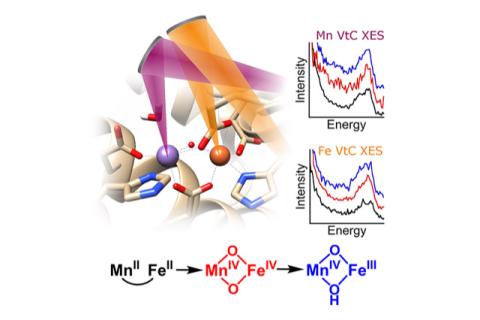
Because this RNR has two different metal ions in its active site that it uses to carry out its reaction—one manganese and one iron ion—it is amenable to being studied using x-ray spectroscopy. Specifically, a technique called valence-to-core x-ray emission spectroscopy (XES) is particularly well-suited for uncovering important information about the enzyme's structure during turnover, though this method had never before been used to study a pathway of enzyme intermediates. Using a new instrument designed and built at CHESS—the dual array valence emission spectrometer or DAVES—both the manganese and iron ions could be analyzed simultaneously, for the first time allowing these sample-hungry experiments to be conducted. By using this unique “two-color” approach, three different states of the enzyme were studied and new information was gained about how the enzyme becomes activated for turnover.
Impact:
This study not only adds to our understanding of how this particular RNR enzyme works, but also demonstrates two-color valence-to-core x-ray emission spectroscopy as a new and powerful technique for investigating enzymes in general.
Collaborators:
Chris Pollock, Ken Finkelstein
CHESS
Ryan Martinie, Beth Blaesi, Carsten Krebs, Marty Bollinger
Penn State
Publication Citation:
Martinie, R.J., Blaesi, E.J., Bollinger Jr., J.M., Krebs, C., Finkelstein, K.D., Pollock, C.J. Two-Color Valence-to-Core X-ray Emission Spectroscopy Tracks Cofactor Protonation State in a Class I Ribonucleotide Reductase., Angewandte Chemie Int. Ed. 2018, 57, 1-6, DOI: doi.org/10.1002/anie.201807366 August 3, 2018
Funding:
C.J.P. and E.J.B. thank the National Institutes of Health for National Research Service Awards (GM113389‐01 and GM116353‐01). This material is based upon work supported by the National Science Foundation Graduate Research Fellowship Program under grant no. DGE1255832 (R.J.M.) and by the National Institutes of Health (GM55365 to J.M.B. and C.K.). This work is based upon research conducted at the Cornell High Energy Synchrotron Source (CHESS) which is supported by the National Science Foundation under award DMR‐1332208.