What did the Scientists Discover?
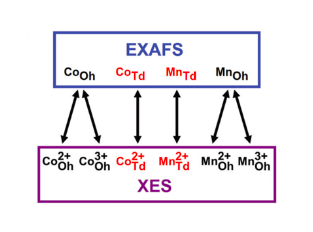
Understanding the distribution of cations in an atomic lattice is important because of their impact on the optical, magnetic, electronic, and catalytic properties. For example, in the spinel cobalt manganese oxide system, depending on synthesis or processing parameters, Co and Mn atoms can shuffle between octahedral and tetrahedral sites in the lattice, and this dramatically affects supercapacitor performance and activity during oxidation-reduction reactions. Current characterization methods have significant limitations in determining complete site occupation information of cations, especially within nanoparticles. Since material properties are exceedingly dependent on electronic structure, it becomes imperative to develop methods to reliably determine oxidation state and site occupation of cations in complex systems.
In this work, we apply x-ray emission spectroscopy (XES) as an accurate and superior method of extracting cation site occupation. We take advantage of spin, ligand geometry, and oxidation state sensitivity of the K-β1,3 (3p - 1s) emission line to study the geometric structure of a model system of spinel phase cobalt manganese oxide nanoparticles with different Co to Mn ratio, contaminated with other impure oxide phases. We focus on nanoparticles since they are actively studied for emerging applications, and demonstrate that from XES we can reliably extract the concentration of cations having different ligand geometries (site occupation) and oxidation states. Furthermore, we compare site occupation of cations obtained using XES to results from extended x-ray absorption fine structure (EXAFS) and demonstrate that while both techniques reach similar findings, XES is superior. It provides not only elemental site occupation, but also oxidation state site occupation of all cation species. Moreover, we show that errors in site occupation for EXAFS can be as high as 35%, whereas for XES, this error is consistently less than 10%.
Impact:
Spinels are an important class of materials with applications for batteries, electrocatalysis and solar cells. By providing an accessible analysis method to determine the structural details of materials, we can predict and thus manipulate properties such as the electrical conductivity which has a direct correlation to performance of various devices.
Collaborators:
Richard D. Robinson, Dept of Materials Science and Eng., Cornell University, E-mail: rdr82@cornell.edu
Anuj Bhargava, Dept of Materials Science and Eng., Cornell University
Cindy Y. Chen, Dept of Materials Science and Eng., Cornell University
Kenneth D. Finkelstein, Cornell High Energy Synchrotron Source (CHESS)
Matthew J. Ward, CLS@APS, Sector 20 Advanced Photon Source, Canadian Light Source Inc., and Cornell High Energy Synchrotron Source (CHESS)
Publication citation:
Bhargava A., Chen C.Y., Finkelstein K.D., Ward M.J., Robinson R.D. "X-ray emission spectroscopy: an effective route to extract site occupation of cations," Phys. Chem. Chem Phys., 2018, Advanced Article, doi:10.1039/C8CP04628J, July 20, 2018.
Funding:
This work was supported in part by the National Science Foundation (NSF) under award number DMR-1809429, DMR-1149036, and CHE-1507753. This work made use of Cornell Center for Materials Research (CCMR) and Cornell High Energy Synchrotron Source (CHESS) facilities, and was funded in part by National Science Foundation under award number DMR-1719875 and DMR-1332208, respectively. CLS@APS facilities (Sector 20-BM) at the Advanced Photon Source (APS) are supported by the U.S. Department of Energy (DOE), NSERC Canada, the University of Washington, the Canadian Light Source (CLS), and the APS. Use of the APS is supported by the DOE under Contract No. DE-AC02-06CH11357. The CLS is financially supported by NSERC Canada, CIHR, NRC, and the University of Saskatchewan.