Tags
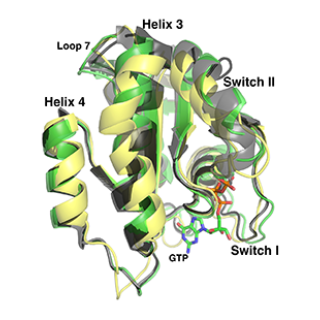
Predicting X-ray solution scattering from flexible macromolecules
Proteins are molecular machines that participate in the vast majority of activities that occur in any living system. To carry out their functions, they undergo dynamic changes in structure and shape and interact with other molecular systems. Characterizing these intramolecular motions provides insight into the molecular basis of protein function and, in turn, can result in a deeper understanding of a vast range of physiological functions.

Workshop on electric circuits spreads a current of enthusiasm from NYS to Puerto Rico
On a cold Saturday morning on October 13, things were heating up in the 7thfloor of Clark Hall. A small group of teachers from all over New York State along with a representative from Puerto Rico, gathered in the Bethe auditorium to learn how to teach electric circuits following the New York State Science Learning Standards. And had a great time while at it!

Hispanic-serving institutions partner with CHESS
Héctor D. Abruña, the Emile M. Chamot Professor in the Department of Chemistry and Chemical Biology, has been working to promote underrepresented minorities in the sciences at Cornell for more than 30 years.
His efforts and hard work are coming to fruition in the form of grant funding from the National Science Foundation, which will help students from his native Puerto Rico access the experimental resources and expertise available to them at the Cornell High Energy Synchrotron Source (CHESS).

Defense spending bill extends Air Force research partnership with CHESS
For the past 10 years, the U.S. Air Force has funded research on high-performance materials at the Cornell High Energy Synchrotron Source (CHESS). The partnership has resulted in numerous advances, including a greater understanding of metal fatigue and analysis of the best metals for aircraft. This partnership was extended with $8 million in funding to CHESS as part of the fiscal year 2019 defense appropriations bill, a $674.4 billion package that President Donald Trump signed into law Oct. 1. The bill passed both the U.S. Senate – supported by New York Sens. Charles Schumer, who is Senate minority leader, and Kirsten Gillibrand – and the U.S. House of Representatives late last month.

50 years Later, Wilson Lab stays cutting edge
This October marks the 50th anniversary of the dedication of the Wilson Synchrotron Laboratory. Initially built for $11million and promising to deliver cutting-edge research in elementary particle physics, it was the NSF’s largest project at that time. Fifty years later, the lab is going through its biggest upgrade in decades.
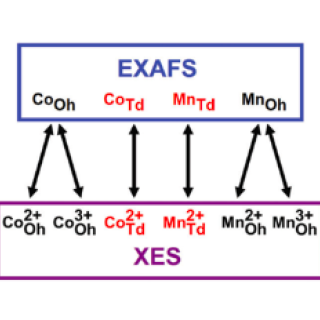
X-ray emission spectroscopy: an effective route to extract site occupation of cations
In crystals, atoms sit at specific locations in the lattice. The arrangement is usually ordered, and properties can be predicted from such arrangements. Sometimes atoms become rearranged, swapping places, and then basic theories for material properties breakdown. In this work, we demonstrate a method to find the position of atoms in a more precise manner than typically used, and one which supplies more information about the atoms as well.
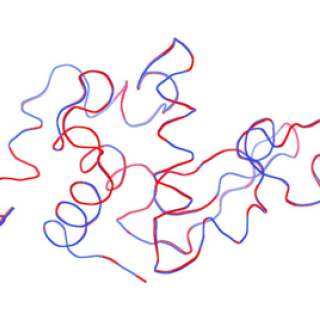
Solving protein structure from sparse serial microcrystal diffraction data at a storage ring synchrotron source
X-ray crystallography allows determination of the atomic structure of proteins, information that is essential to understanding the proteins. It was thought that there was a minimum size of crystal that could be used at storage ring x-ray sources. This paper shows that the minimum size barrier can be overcome.
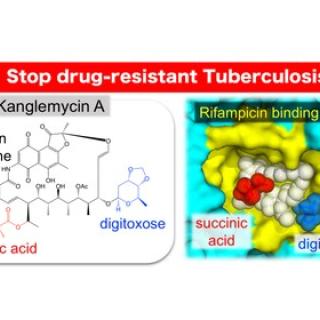
A naturally occurring antibiotic active against drug-resistant tuberculosis
A naturally occurring antibiotic called kanglemycin A is effective against Mycobacterium tuberculosis, the bacteria that cause tuberculosis, even in drug-resistant strains, according to an international team of researchers who used chemistry, molecular biology, microbiology, and X-ray crystallography to show how the compound maintains its activity.





